About
Pneumonia is the world’s leading infectious disease killer of children under five, accounting for more than 800,000 – or around 15% of child deaths annually. Nearly half of all these deaths occur in sub-Saharan Africa. If children are diagnosed early and correctly, pneumonia can be easily treated with affordable oral antibiotics. Severe cases can be referred to facilities that are better equipped to deliver advanced care. However, inaccurate diagnosis, inadequate supplies of medicines and weak referral systems remain a challenge in fighting the disease in low- and middle-income countries and in humanitarian contexts.
WHO guidelines
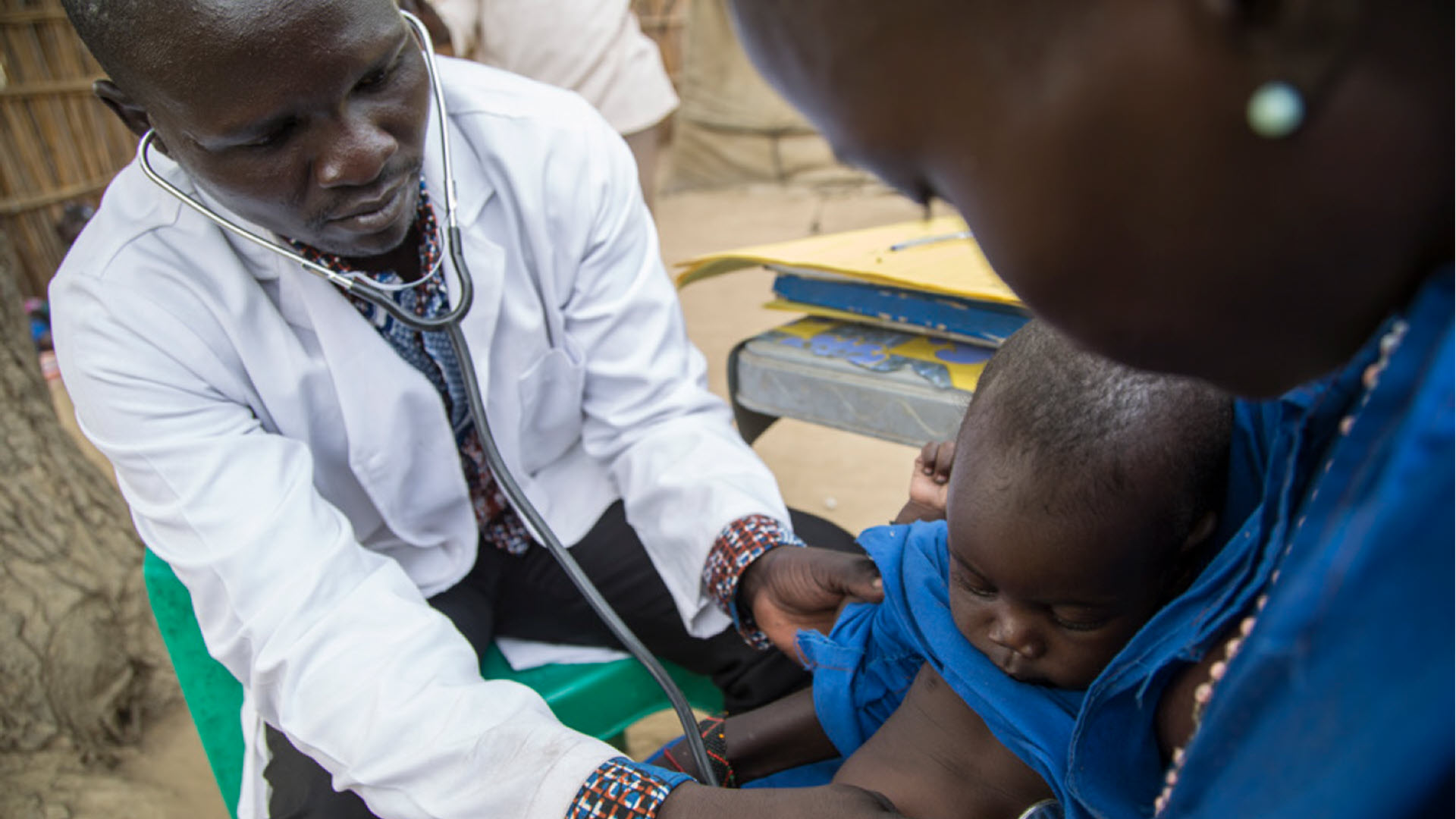
According to the World Health Organization (WHO) international guidelines for the management of pneumonia, assessment of a child’s respiratory rate is a critical component for diagnosing children with pneumonia in low-resource settings. However, counting respiratory rates is challenging, particularly in children as they may breathe irregularly, and it can be difficult to keep them calm for an entire minute. Miscounting by community health workers and even health providers is common, which can lead to inaccurate diagnosis and treatment.
The Philips Children’s Automated Respiration Monitor (ChARM) was developed in response to this challenge. The ChARM device automatically measures the respiratory rate of a child and classifies the breathing rate according to the WHO guidelines for childhood pneumonia.
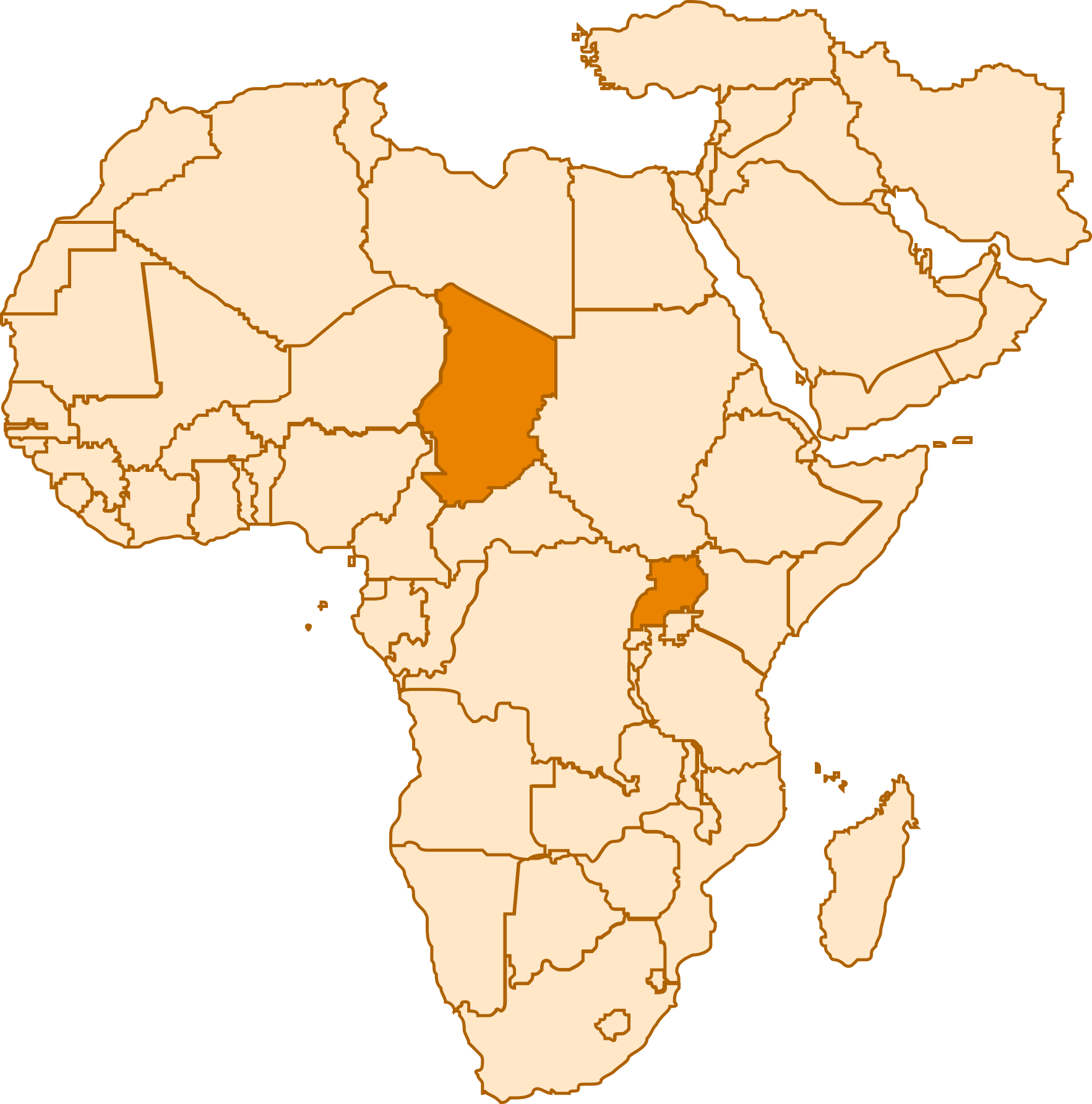
Complementing earlier studies of ChARM, this project specifically evaluated the effectiveness of ChARM by community health workers in low literacy refugee settings in Chad and Uganda.